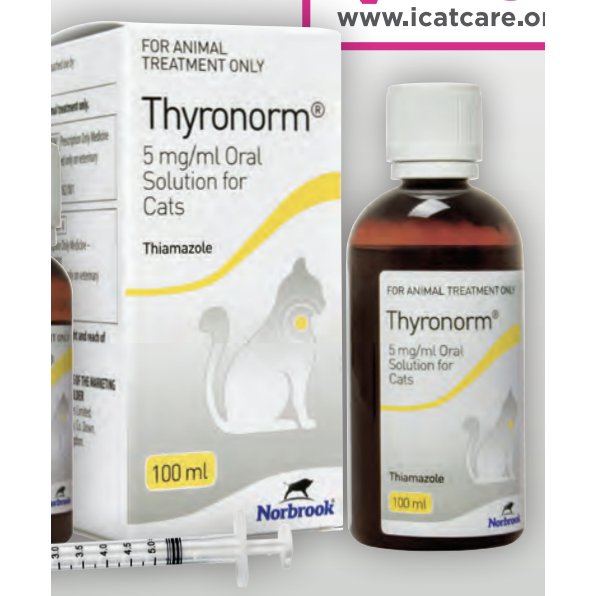
Thyronorm 5 mg/ml Oral Solution for Cats , POM-V
Product Description
Description
Indications for use, specifying the target species
For the stabilisation of hyperthyroidism in cats prior to surgical thyroidectomy.
For the long term treatment of feline hyperthyroidism.
Contraindications
Do not use in cats suffering from systemic disease such as primary liver disease or diabetes mellitus.
Do not use in cats showing signs of autoimmune disease.
Do not use in animals with disorders of white blood cells, such as neutropenia and lymphopenia.
Do not use in animals with platelet disorders and coagulopathies (particularly thrombocytopenia).
Do not use in pregnant or lactating females.
Please refer to section, “Use during pregnancy, lactation or lay”
Do not use in case of hypersensitivity to the active substance or to any of the excipients.
Special warnings for each target species
In order to enhance stabilisation of the hyperthyroid patient the same feeding and dosing schedule should be used daily.
Special precautions for use
Special precautions for use in animals
If more than 10 mg per day is required animals should be monitored particularly carefully.
Use of the product in cats with renal dysfunction should be subject to careful risk:benefit assessment by the clinician. Due to the effect thiamazole can have on reducing the glomerular filtration rate, the effect of therapy on renal function should be monitored closely as deterioration of an underlying condition may occur.
Haematology must be monitored due to risk of leucopenia or haemolytic anaemia before initiating treatment and closely afterwards.
Any animal that suddenly appears unwell during therapy, particularly if they are febrile, should have a blood sample taken for routine haematology and biochemistry. Neutropenic animals (neutrophil counts <2.5 x 109/l) should be treated with prophylactic bactericidal antibacterial drugs and supportive therapy.
Please refer to section, “Amounts to be administered and administration route” for monitoring instructions.
As thiamazole can cause haemoconcentration, cats should always have access to drinking water.
In hyperthyroid cats, gastrointestinal disorders are common and may interfere with the success of the oral therapy.
Special precautions to be taken by the person administering the veterinary medicinal product to animals
People with known hypersensitivity (allergy) to thiamazole or one of the excipients should avoid contact with the veterinary medicinal product. If allergic symptoms develop, such as a skin rash, swelling of the face, lips or eyes or difficulty in breathing, you should seek medical attention immediately and show the package leaflet or label to the doctor.
This product may cause skin or eye irritation. Avoid eye contact including hand to eye contact. In case of accidental eye contact, rinse eyes immediately with clean running water. If irritation develops, seek medical advice.
Wash hands with soap and water after administration of the product and handling the vomit of or litter used by treated animals. Wash any spillages or spatter from skin immediately. Thiamazole may cause gastrointestinal disturbances, headache, fever, joint pain, pruritus (itching) and pancytopaenia (decrease in blood cells and platelets).
Avoid dermal and oral exposure, including hand-to-mouth contact.
Do not eat, drink or smoke while handling the product or used litter.
In case of accidental ingestion, seek medical advice immediately and show the package leaflet or the label to the physician
Following administration of the product any residual product remaining on the tip of the dosing syringe should be wiped clean with a tissue. The contaminated tissue should be immediately disposed of.
The used syringe should be stored with the product in the original carton.
As thiamazole is a suspected human teratogen, women of child-bearing age must wear non-permeable single use gloves when administering the product or handling the litter/vomit of treated cats.
If you are pregnant, think you may be pregnant or are attempting to conceive, you should not administer the product or handle the litter/vomit of treated cats.
Collect in Store
This item is available for collection.
Loyalty Scheme
Earn up to 60 loyalty points with this product.



Share
Thyronorm 5 mg/ml Oral Solution for Cats , POM-V
Facebook Twitter Email Pinterest Telegram